Description
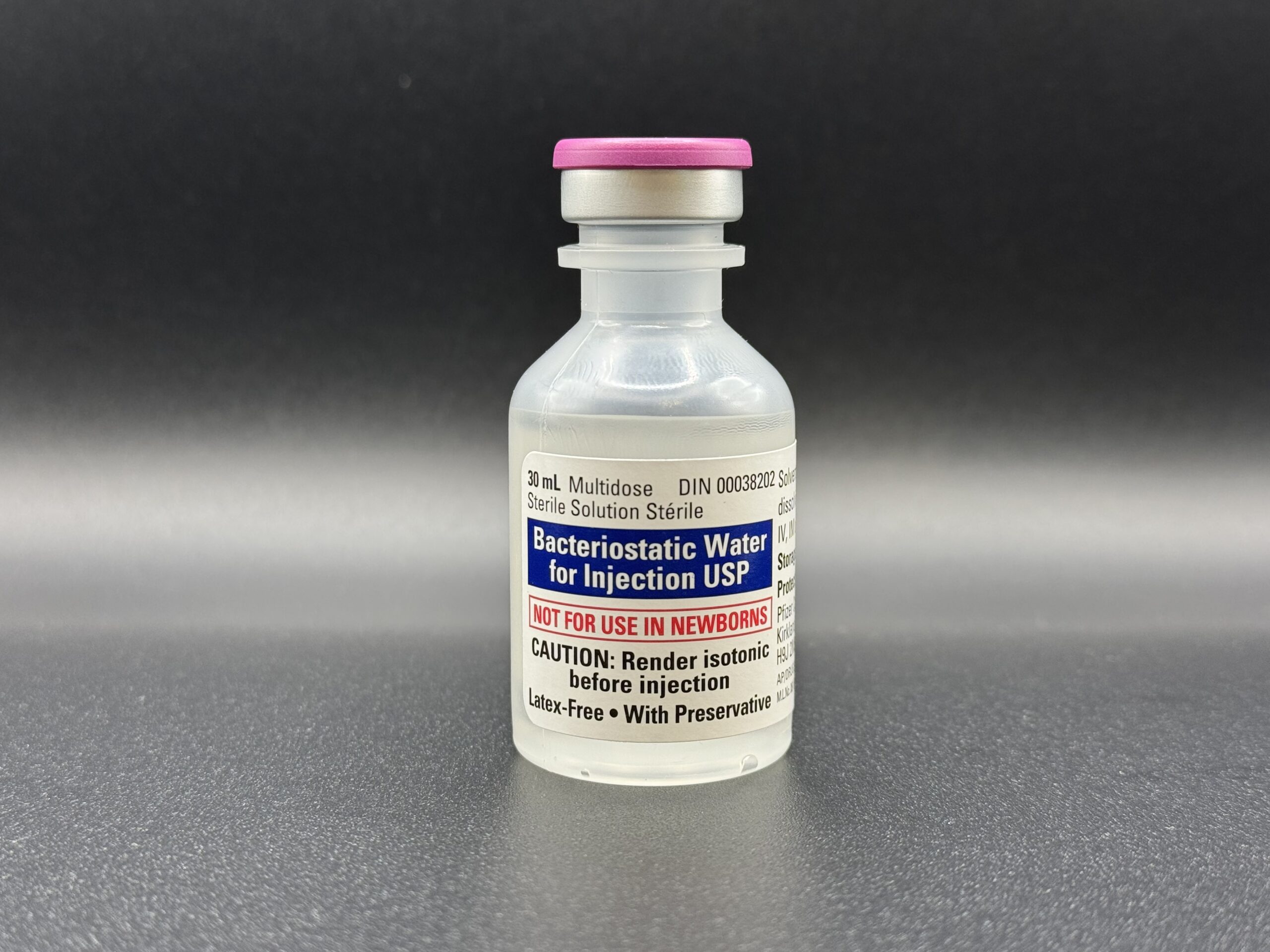
Hospira Bacteriostatic Water 30ml
Premium Grade | Multi-Dose Research Solvent
As a primary choice for laboratory researchers in Canada, this multi-dose vial allows for repeated withdrawals under sterile conditions. It is specifically designed for the reconstitution of research peptides and other compounds, ensuring the stabilization of lyophilized proteins for up to 28 days post-puncture.
Scientific Background & Antimicrobial Efficacy:
In the field of molecular biology, the integrity of research solutes depends heavily on the prevention of microbial contamination. Hospira Bacteriostatic Water 30ml utilizes benzyl alcohol to effectively inhibit the proliferation of most bacteria, including common Gram-positive and Gram-negative strains.
Unlike “bactericidal” agents that kill bacteria instantly, a “bacteriostatic” agent like the one used in this Pfizer-Hospira formula works by arresting the metabolic growth of microorganisms. This distinction is vital for researchers who need to maintain a pristine chemical environment over several weeks of testing.
Reconstitution & Stability Optimization:
The precise pH of 5.7 in Hospira Bacteriostatic Water is formulated to support the structural stability of delicate amino acid chains. Many research peptides are highly sensitive to pH fluctuations. Using a USP-grade solvent ensures that your lyophilized compounds do not degrade prematurely through oxidation or hydrolysis.
Furthermore, the USP designation guarantees that the water has undergone rigorous testing for total organic carbon and conductivity. This meets the strictest laboratory purity standards required for high-stakes cellular research.
Safety Protocols and Disposal:
Proper laboratory handling is mandatory when working with Hospira Bacteriostatic Water 30ml. Researchers should always utilize a new, sterile needle and syringe for each withdrawal to minimize the risk of cross-contamination. While the benzyl alcohol preservative is robust, its efficacy diminishes after 28 days following the initial puncture.
Laboratories must adhere to strict disposal guidelines for biohazardous waste. It is also important to note that benzyl alcohol is incompatible with certain sensitive biological tissues. This is why this product is strictly labeled for in-vitro laboratory research and not for any clinical application. This product is for in-vitro research.
Explore our full range of ancillary research products.
Technical Specifications
| Product Name | Hospira Bacteriostatic Water 30ml (bac water) |
|---|---|
| Manufacturer | Hospira (Pfizer Canada) |
| Active Ingredient | Benzyl Alcohol 0.9% |
| Pharmacopeia Grade | USP (United States Pharmacopeia) |
| DIN | 00038202 |
| Storage Temperature | 20°C to 25°C (68°F to 77°F) |
| Volume / Format | 30 mL / Multi-dose plastic vial |